Ines Lacarne
This is a summary of a paper by researchers in the laboratory of ICORD researcher Dr. Stephanie Willerth.
Dr. Willerth was recently named one of the 2015 Young Innovators in Cellular and Molecular Bioengineering by the Biomedical Engineering Society based on the work presented in this paper. This work was supported by an ICORD seed grant.
Original article: Gomez, J., Edgar, J., Agbay, A., Bibault, E., Montgomery, A., Mohtaram, N., & Willerth, S. (2015). Incorporation of Retinoic Acid Releasing Microspheres into Pluripotent Stem Cell Aggregates for Inducing Neuronal Differentiation. Cel. Mol. Bioeng. You can find the original article here.
Introduction
A stem cell is a type of cell that can turn into (or “differentiate”) into a number of different specialized cell types by means of internal or external triggers, such as a cell’s genes, or chemicals or molecules in the cell’s environment. (Learn more about stem cells here).
Stem cell technology can give us the ability to engineer human cells in order to replace and repair lost or damaged cells. The use of stem cells promises to be a revolutionary tool for regenerative medicine as it offers a large number of potential applications. Currently, only a few forms of stem cell therapy are successfully being done, but research is ongoing to develop treatments for neurodegenerative disorders and other diseases.
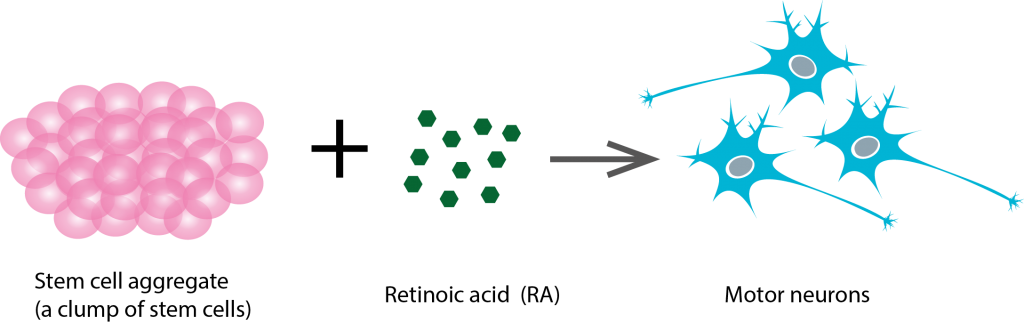
This experiments by the Willerth lab looked at generating neural stem cells. Neural stem cells are responsible for the development of neurons and other cells of the nervous system. The aim of the experiment was to see whether cultured neural stem cells could be differentiated into neurons. Those neurons could be then transplanted to the injury site, such as a lesion in the brain or the injured spinal cord.
Retinoic acid (RA) molecules have been previously shown to trigger differentiation of pluripotent stem cells into neurons. RA is a morphogen (a substance that controls the way in which tissue develops to define an organism’s shape and structural features.
In the past, morphogens like RA have been administered in the form of a solution. However, this method has some downsides. Cell consumption rates, cell size and density have previously been shown to be important factors for differentiation of pluripotent stem cells. If the consumption rate of molecules being delivered is too high by the cells on the outside of an aggregate of stem cells, this can lead to low concentrations inside of the aggregate.
Taking these factors into consideration, the researchers decided to test a different method of delivery. RA molecules would be contained within microspheres (tiny spheres made from synthetic materials), which would allow for sustained, uniform delivery into the targeted stem cells.
The use of drug-releasing microspheres into induced pluripotent stem cell aggregates has not been studied in the past. Because microspheres allow for localized delivery of molecules into the cells, the researchers hypothesized that it would be a viable strategy to overcome specific concerns that come with complications of neurological disorders (for example, in the acute phase of SCI, the body’s inflammatory response protects surviving cells but can also compromise survival of transplanted neural stem cells).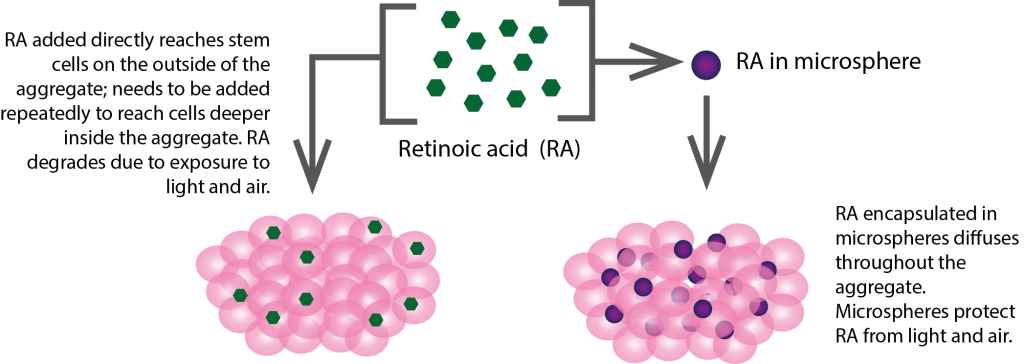
The experiment
First, researchers created microspheres using a polymer (a synthetic chain of molecules) chosen for its low cost, biodegradability, and long lasting properties, since the process of neuronal differentiation can take a long time. The RA molecules were contained within the microspheres. They then took some measurements of the average size of the microspheres they were going to use, as well as the average concentration of RA within the microspheres, since previous research had identified those factors to be important. The microspheres were then incorporated into the stem cell culture. After 5 days, they did not observe any toxic effects.
What did they find?
The researchers took measurements to assess the effects of the RA. They assessed the ability of the stem cells to differentiate into various cell types, and found that they were less able to do so, meaning they were becoming specialized. This suggested that the incorporation of RA had had the desired effect. After 7 days of culture, they observed that the cell clusters containing the RA microspheres had successfully differentiated into neurons.
The researchers found that the use of microspheres allowed for more homogeneous delivery of RA in the cells compared to techniques used in previous research. By comparing different sizes of microspheres in the experiment, they identified a specific diameter of microspheres that was most efficiently incorporated into stem cell aggregates.
What does this mean?
The results of this study suggest that the use of microspheres is a viable alternative for delivering morphogens, which is promising. If RA-releasing microspheres can be used to differentiate pluripotent stem cells into neurons, a future experiment could be done over a longer period of time to see if it’s possible to differentiate pluripotent stem cells into other types of neurons, such as motor neurons.
To learn more about what’s going on in Dr. Willerth’s laboratory, check out the Winter 2015 issue of the ICORDian Community Newsletter where we profile masters student Jose Gomez and his work on microspheres.